Imbalance of the autonomic nervous system (sympathetic/parasympathetic) is known to contribute to the pathophysiology of multiple cardiovascular diseases, including AF, MI (and related ventricular arrhythmias) and heart failure. The concept of neuro-immune axis has been proposed to tightly integrate the brain–heart–periphery axis, which is characterised by various pathways of the anti-inflammatory properties of the vagus nerve.1,2 For instance, immune responses can activate the hypothalamic–pituitary–adrenal axis through the vagal afferents and the cholinergic anti-inflammatory pathway through the vago-parasympathetic and sympathetic reflexes.3
Modulating this neuro-immune axis could play a key role in the alleviation of several cardiovascular diseases. Several modalities have been developed to modulate this axis and restore a favourable balance between the sympathetic and parasympathetic system. A few examples of this include direct vagus nerve stimulation (VNS), baroreceptor stimulation, renal artery sympathetic denervation, low-level tragus stimulation (LLTS), cardiac sympathetic denervation and spinal cord stimulation.4 Among them, direct cervical VNS has been used in multiple diseases, including epilepsy, drug-resistant depression, migraine, angina pectoris, hypertension and impaired upper limb function after stroke.5–11 In patients with heart failure with reduced ejection fraction (HFrEF), three major clinical trials of direct VNS have yielded mixed results.12–14 One of the major drawbacks of cervical vagus stimulation is the invasive nature of this treatment with inherent surgical complications and poor patient tolerance.15
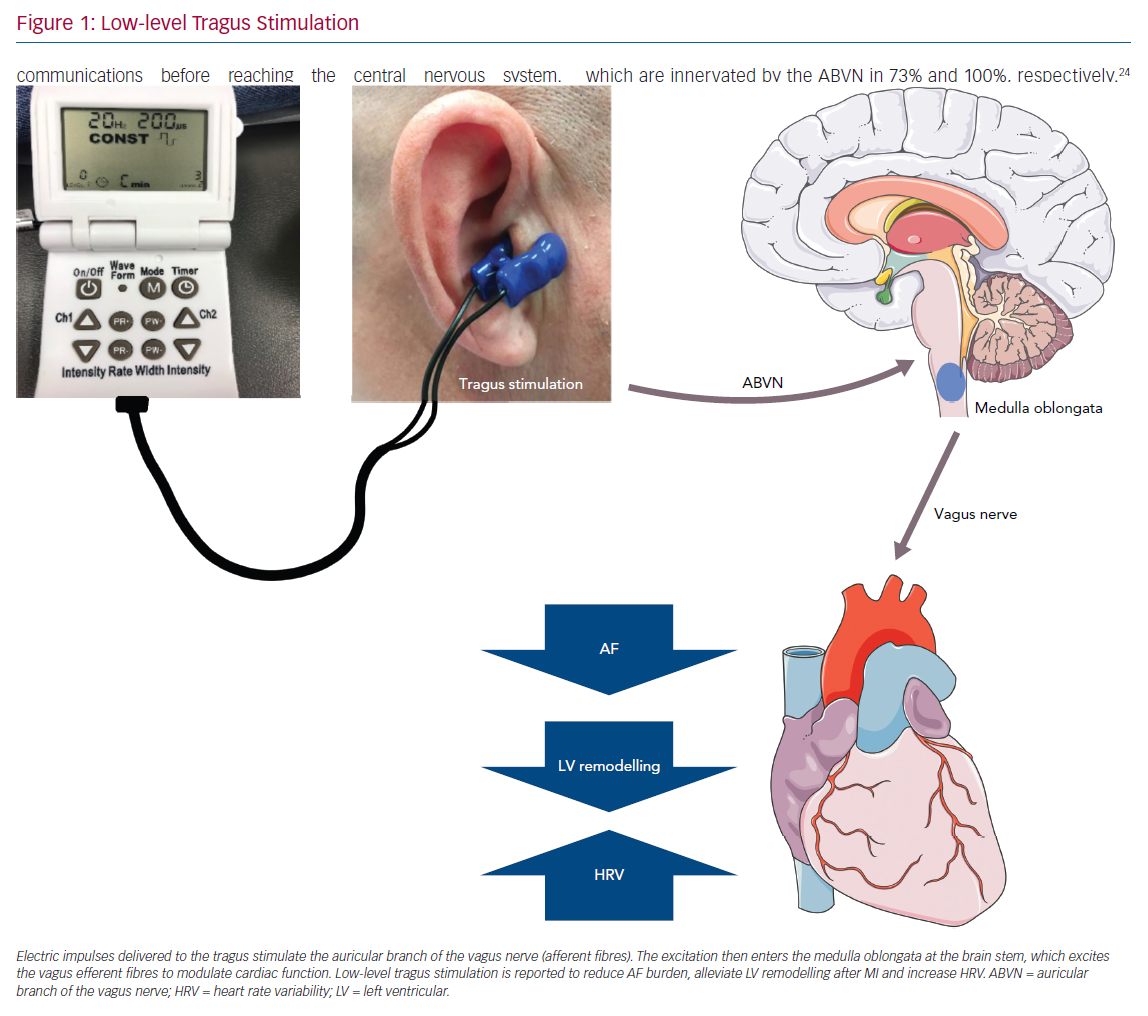
Designed to stimulate the auricular branch of the vagus nerve (ABVN), LLTS is a non-invasive transcutaneous technique of VNS that can modulate the autonomic function (Figure 1).16 The aim of this current review is to summarise the evidence to date pertaining to the use of LLTS in various cardiovascular disease states.
The Anatomical and Physiological Basis of Low-level Tragus Stimulation
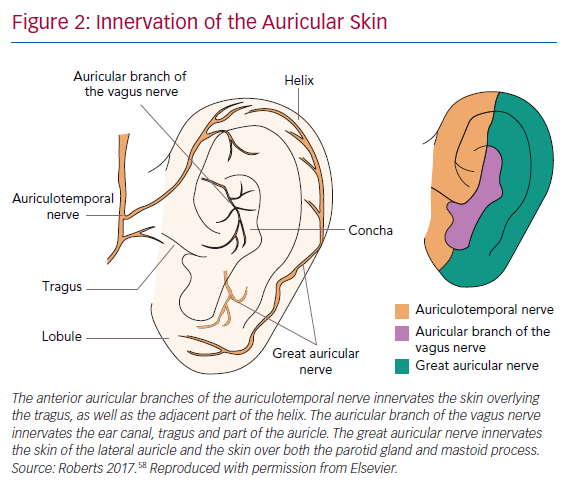
Twenty per cent of the vagus nerve fibres are efferent fibres originating from the brainstem, providing parasympathetic control of the viscera, peripheral vasculature and heart. The remaining 80% of the nerve fibres are afferent fibres that relay sensory information from the periphery to the central nervous system. The afferent fibres distribute widely in the heart, lungs, liver, adrenal medulla and the gastrointestinal tract up to the splenic flexure of the colon.17 The vagus nerve contains three fibre types: highly myelinated A fibres, which have low activation thresholds; lightly myelinated B fibres; and unmyelinated C fibres, which have high activation thresholds.18,19 The ABVN is the cutaneous branch of the vagus nerve which innervates the antihelix, tragus and cavity of the concha.20 The innervation of the auricle is shown in Figure 2. Currently, how the external auricle is innervated remains controversial. In an anatomical study, ABVN was able to be observed in 94% of cases (17/18). All of the ABVN were observed to distribute to the external acoustic meatus and auricle. Cutaneous branches from the facial nerve were observed to innervate the external acoustic meatus as well.21 The nerve branches around the auricle and the acoustic meatus have various complex communications before reaching the central nervous system. Nevertheless, ample evidence indicates that the ABVN remains the most important innervation of the external auricle and the cymba conchae.22
Brain activation patterns, based on functional MRI (fMRI) during LLTS, were associated with greater activation of the vagal centres (dorsal vagal complex) throughout cortical, subcortical and cerebellar brain regions compared to sham stimulation.23 In one of the most cited anatomical studies, 14 ears from seven cadavers were examined. A total of 45% of the ears were shown to be innervated by the great auricular nerve (GAN), 9% by the auriculotemporal nerve (ATN) and 46% were innervated by both the GAN and ATN. However, there has been some debate on the optimal site of transcutaneous VNS (tVNS). An fMRI study recently showed that stimulation at the cymba conchae had a greater effect on the vagal pathways and the dorsal vagal complex compared to three other locations: the inner surface of the tragus, the posterior-inferior wall of the external acoustic meatus and the earlobe.23 A recent study used the triangular fossa innervated by the GAN as the stimulation site, because it allowed stimulation for a longer period of time without interfering with patient comfort, daily hygiene requirements and sleep. Most importantly, it is located proximal to the antihelix and the concha, which are innervated by the ABVN in 73% and 100%, respectively.24 Stimulating the effective triangular fossa was effective in suppressing postoperative AF in this study.
Parameters of Low-level Tragus Stimulation
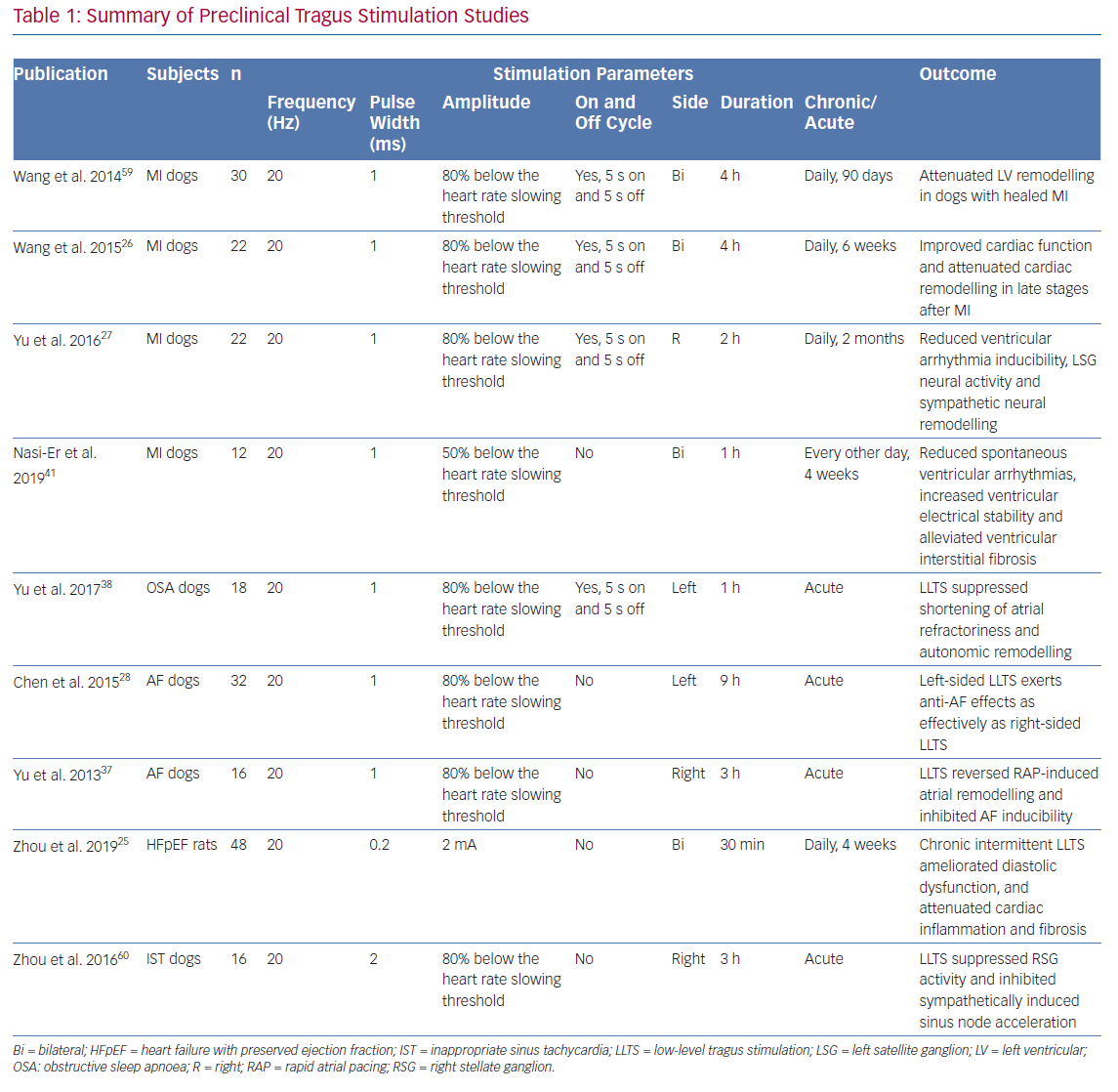
Currently, optimal settings of LLTS remain undetermined. Parameters of LLTS for both preclinical and clinical studies have generally been empirical (Tables 1 and 2). In a rat model of heart failure with preserved ejection fraction (HFpEF), LLTS below the threshold of heart rate reduction was applied by placing two oppositely charged magnetic electrodes over the auricular concha region at 20 Hz frequency, 0.2 ms pulse duration and 2 mA amplitude.25 In a rat MI model, LLTS at 20 Hz, 1 ms pulse wide and 80% below the threshold of slowing the sinus heart rate was used.26 The same settings were extended to a canine model of post-infarction ventricular arrhythmia.27 The majority of LLTS studies were right-sided, while one canine model study demonstrated that left-sided LLTS is also effective in suppressing AF.28
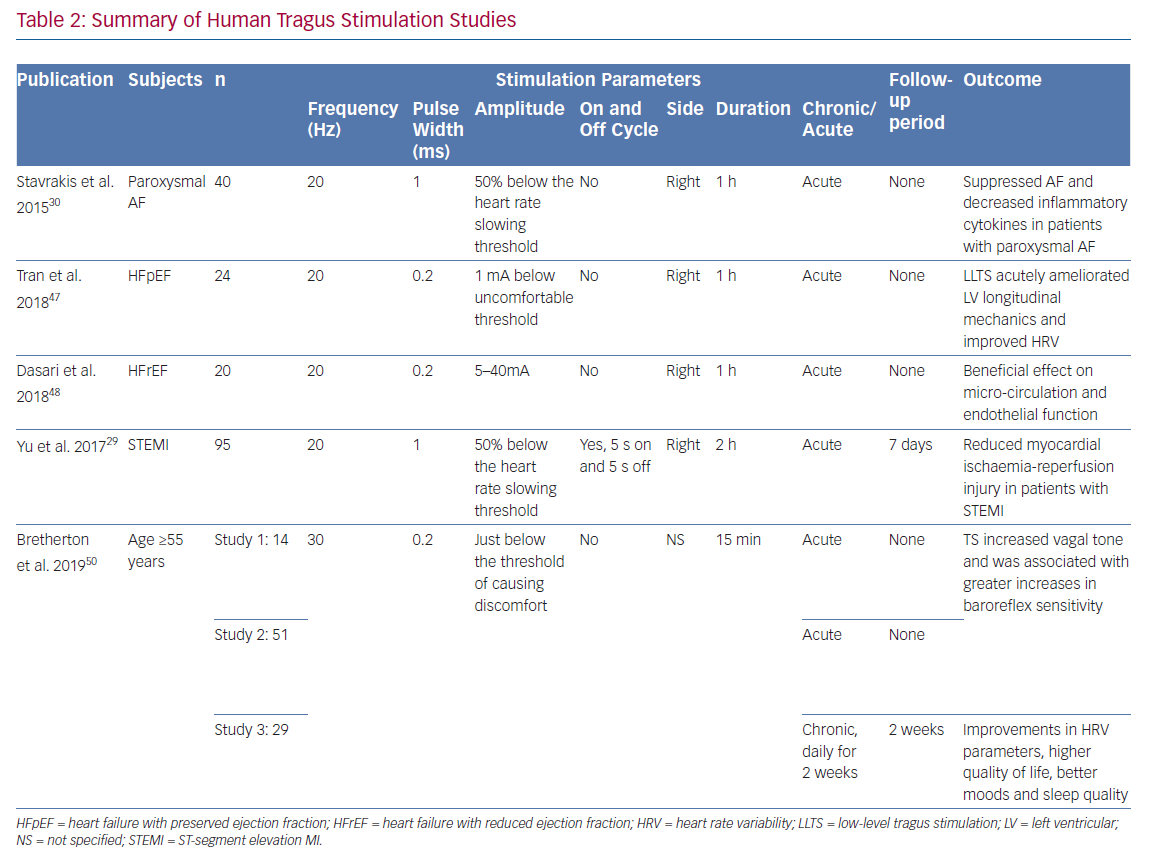
In a clinical study in patients with ST-segment elevation MI (STEMI), LLTS was set at 20 Hz, 1 ms pulse wide and 50% below the heart rate-slowing threshold with a duty cycle of 5 seconds on and 5 seconds off.29 The same parameters were used in an AF suppression study, but in a continuous manner without the on-and-off cycle.30 To test if LLTS has a parameter-specific effect on heart rate, an exploratory study compared nine combinations of stimulating parameters (pulse width: 100 μs, 200 μs, 500 μs; frequency: 1 Hz, 10 Hz, 25 Hz) among 15 healthy volunteers.31 Essentially, the setting of 500 μs and 10 Hz had the strongest effect on heart rate reduction (−2.40 ± 0.275 BPM) during the 60 seconds of stimulation. Recently, a large clinical trial using VNS in patients with HFrEF failed to show a beneficial effect while another trial in patients with HFrEF using different stimulation parameters demonstrated efficacy, highlighting the importance of optimising stimulation parameters.13,32 Mechanistic studies have suggested that the optimal stimulation parameters for VNS are at the point at which the afferent and efferent fibres are activated in a well-balanced manner, i.e. the afferent-driven decreases in central parasympathetic drive are counteracted by direct activation of the cardiac parasympathetic efferent projections to the intrinsic cardiac autonomic nervous system, resulting in a relatively neutral heart rate response.33 Consistent with this notion, a recent study of LLTS (30 Hz,10–50 mA, 200 ms, which was slightly below the sensory threshold) in healthy volunteers showed a significant decrease in muscle sympathetic nerve activity (MSNA) without a change in heart rate.34 Recently, the works by Sclocco et al. suggested that tVNS is headed toward a more refined and sophisticated direction.35,36 Through respiratory gating and EEG-informed fMRI, the efficacy and accuracy of this therapy can be further improved.
Results in Animal Models
AF
It has been established that the abnormal regulation of the cardiac autonomic nervous system contributes to the initiation and perpetuation of AF. High sympathetic/vagal tone contribute to the substrate of AF while sudden changes in autonomic balance lead to AF episodes. Theoretically, suppressing either the sympathetic or parasympathetic tone could have a moderating effect on AF. There have been at least two animal models testing the acute effect of LLTS. For example, in a canine AF model, right sided LLTS reduced AF inducibility and reversed acute atrial electrical remodelling. Its anti-remodelling effect was indicated by a prolonged effective refractory period and a narrowed window of vulnerability (to measure the difficulty of AF induction).37 In a canine model of obstructive sleep apnoea, LLTS suppressed shortening of atrial refractoriness and autonomic remodelling caused by obstructive sleep apnoea, thus reducing the vulnerability to AF.38
Post MI
The close relationship between ventricular arrhythmias after MI and sympathetic activation is well recognised.39,40 Suppressing the sympathetic tone and increasing the parasympathetic tone protected against post-MI ventricular arrhythmias.
In a canine MI model, right-sided LLTS performed 2 hours a day for 2 months reduced left stellate ganglion activity in both frequency and amplitude.27 Both of these parameters of neural activity were significantly lower in the MI plus LLTS group than the MI group alone. Ventricular arrhythmias inducibility evaluated by ventricular programed stimulation was also suppressed by LLTS. Besides electrical remodelling, in a canine model intermittent bilateral LLTS for 4 weeks alleviated left ventricular remodelling by reducing ventricular interstitial fibrosis post MI.41 In another post-MI canine model, 6 weeks of bilateral LLTS (20 Hz, 1 ms pulse wide, 80% below the sinus slowing threshold) for 4 hours per day reduced left ventricle dilation and infarct size, improved both left ventricular contractility and diastolic function and alleviated myocardial fibrosis.26
Heart Failure with Preserved Ejection Fraction
HFpEF, another clinical syndrome associated with dysregulation of the autonomic nervous system and inflammation, comprises approximately 50% of all patients with heart failure.42 Patients with HFpEF have been shown to have increases in plasma renin activity and arginine vasopressin compared with healthy controls.43 In a rat model of HFpEF (Dahl salt-sensitive rats), LLTS (20 Hz, 2 mA, 0.2 ms) attenuated the elevation of both systolic and diastolic blood pressure. Echocardiography revealed that LLTS improved left ventricular hypertrophy, circumferential strain and diastolic function as measured by E/A ratio and E/e’ ratio.25 Histological studies showed that LLTS attenuated ventricular inflammatory cell infiltration and fibrosis. Downregulation of the pro-inflammatory and pro-fibrotic genes (interleukin [IL]-11, IL18, IL23-A and osteopontin) was seen in the LLTS group as well.25
Mechanisms of Low-level Tragus Stimulation
Tragus stimulation exerts its modulating effects through an integrated nervous reflex system. The ABVN is the afferent nerve fibre while the medulla oblongata (nuclei including NTS, nucleus of the solitary tract; NSNT, nucleus spinalis of the trigeminal nerve; NA, nucleus ambiguous; DMN, dorsal motor nucleus) function as the reflex centre.44 The efferent vagus nerve then arrives at the ganglionated plexi within the epicardial fat pads to achieve the related physiological effects. Though the neuropathway of tVNS is grossly understood, the exact mechanisms underlying the various beneficial effects of LLTS are still poorly understood. The inherent sympathovagal imbalance is different in various cardiovascular states. Modulation of this tone may have variable beneficial effects and is likely directly related to the underlying pathophysiology. For example, even in heart failure, acute and chronic states exhibit a wide spectrum of sympathovagal imbalance and there are likely differential effects of non-invasive vagal modulation. In common cardiovascular disease states, vagal stimulation probably suppress arrhythmias, ischaemia and heart failure through different mechanisms. Up-regulation of c-fos (a proto-oncogene expressed within some neurons following depolarisation) and nerve growth factor (NGF) expression in left superior ganglionated plexus and the left stellate ganglion is found to contribute to autonomic remodelling of AF. LLTS suppressed AF by down-regulating both c-fos and NGF.38 Connexins (Cx) are an essential component of gap junctions and a key player in the formation of AF substrate. LLTS has been shown to suppress AF by preventing the loss of atrial Cxs (Cx40 and Cx43).28
At the cardiac remodelling and fibrosis level, downregulation of matrix metalloproteinase 9 and transforming growth factor-beta 1 was observed in LLTS treated post-MI dogs.26 Small conductance calcium-activated potassium channel type 2 (SK2) is an ion channel molecule responsible for after-hyperpolarisation that suppresses nerve discharges that are crucial to regulating neuronal excitability. In a canine MI model, LLTS down-regulated the NGF protein and up-regulated the SK2 protein.27 These studies indicate that LLTS affects the pathophysiological progress on multiple levels – from neural remodelling to myocardial remodelling.
Human Studies
AF
Reports of tragus stimulation on human subjects are still limited. LLTS for 1 hour during ablation procedures reduced the duration of induced AF, while levels of inflammatory markers (circulatory tumour necrosis factor [TNF] alpha and C-reactive protein) were reduced.30 In patients with paroxysmal AF who received chronic LLTS for 1 h/day for 6 months, there was a 85% decrease in AF burden in the active stimulation group compared to the sham group (stimulating the earlobe).45
MI and Angina
In patients with STEMI undergoing primary percutaneous coronary intervention, 2 hours of right LLTS after reperfusion attenuated reperfusion-related ventricular arrhythmias during the first 24 hours, with smaller area under the curve of creatine kinase myocardial band, which theoretically indicated reduced infarct size.29 In addition to reduced ventricular arrhythmias and cardiac biomarkers, significant improvement in left ventricular ejection fraction and N-terminal pro–B-type natriuretic peptide was observed in the LLTS group. The wall motion index, calculated using the 16-segment model, was used to evaluate the extent of left ventricular dysfunction. The percentage of left ventricular akinetic and dyskinetic segments was significantly lower in the LLTS group than the sham group. The level of inflammatory cytokines including IL-6, IL1-beta, TNF-alpha and HMGB1 were significantly reduced. However, MRI was not conducted to accurately evaluate the infarct size and their differences between groups. It remains to be proven if chronic LLTS would improve morbidity and mortality in patients with STEMI.
Alongside STEMI, the application of vagal neural stimulation for the treatment of coronary artery disease is also worth mentioning. Though conducted in a small population of patients, the clinical and histological messages in the article by Zamotrinsky et al. were impressive.46 Vagal stimulation via trans-auricular electroacupuncture showed antianginal effect that could last for 2–3 weeks after the completion of the treatments. The effects of LLTS in refractory angina deserves further study.
Heart Failure
Human studies of LLTS on heart failure patients are also limited. In a prospective, randomised, double-blind, 2×2 cross-over study, 1 hour of LLTS acutely ameliorated left ventricular longitudinal mechanics and favourably altered heart rate variability (HRV) frequency domain components in patients with HFrEF.47 In a pilot, sham controlled, randomised study in patients with HFrEF, 1 hour of LLTS (20 Hz frequency and 200 μs pulse width) led to improvements in microcirculation (as assessed by flow mediated vasodilatation)
and nail bed microcirculation (assessed by laser speckle
contrast imaging).48
Ageing
As people age, the contribution of the parasympathetic activity to the heart declines, whereas sympathetic tone increases, resulting in a less balanced autonomic function. Tragus stimulation was found to be beneficial in healthy young subjects as demonstrated by an increase in HRV.49 After preliminary LLTS studies showed its safety and efficacy as an alternative to invasive stimulation of cervical vagus nerve, Bretherton et al. applied daily LLTS at a level just below the threshold of causing discomfort (usually 2–4 mA) to people ≥55 years of age.50 Higher baroreflex sensitivity and HRV were observed by LLTS. Furthermore, it was reported in the same article that in 26 healthy adults with an average age of 64 years, daily sessions of LLTS for 14 days resulted in improvements in HRV parameters, higher quality of life questionnaire score (SF-36), better moods (measured by profile of mood states questionnaire) and better sleep quality (measured by sleep questionnaire), thus showing potential against aging.
Safety of Low-level Tragus Stimulation
The non-invasive nature of tVNS granted it an impressive safety record. Drop in blood pressure and/or heart rate during stimulation is rarely observed. Some mild skin lesions were observed in the study by Stavrakis et al. and they did not recur after adjusting the tension of the medal clips delivering the stimulation.30
A systematic review published by Redgrave et al. summarised all available literature on tVNS, not limited to cardiovascular diseases and found that local irritation (tingling/pain/redness/itching) is the most common discomfort (16.7%) that is attributable to stimulation, with headache being the second (3.3%) and nasopharyngitis (1.6%) the third.51 Dizziness and syncope was found in 20 patients across eight studies, with a prevalence of 1.4%. However, only three severe adverse events were considered tVNS-related.
Conventional wisdom suggests that stimulation of the right vagus nerve exerts more prominent cardiovascular effects than stimulation of the left vagus nerve. Left vagal stimulation is therefore the preferred approach to treat epilepsy.52 This conventional wisdom has been challenged by reported side effects of left VNS studies.53,54 For non-invasive VNS, whether left and right tragus stimulation will affect the sinus node and atrioventricular node is still unknown.
Next Steps
There are some limitations associated with previous studies. These include lack of a well controlled sham group, absence of longitudinal data and small sample sizes. Optimal parameters for stimulation are yet to be determined. Furthermore, the lack of a uniform stimulation parameter makes results less generalisable. Some studies used active stimulation at another site while others used sham stimulation at the same site. All of the current clinical studies are acute or short-term studies except one recent study by Stavrakis et al. reporting that chronic LLTS suppressed paroxysmal AF.45 Longitudinal data are warranted to evaluate the long-term benefit of LLTS.
Lastly, there is a lack of a reliable biomarker to indicate the effectiveness of LLTS. MSNA may serve as an attractive method to determine changes in sympathetic tone with LLTS. However, this technique is time-consuming and lack of widespread availability limits its usage. Serum/plasma markers of neuroendocrine activity (such as vasostatin and catestatin) could also potentially be used to gauge change in neuro endocrine function, pending future data. Stimulus-driven event-related potentials and vagus-sensory evoked potentials in electroencephalogram, fMRI and HRV are potential biomarkers as well.34,55–57 In some clinical studies, a simple reduction in heart rate is considered a marker of parasympathetic activation.29 Individual disease states may have respective biomarkers that would predict adequate response to LLTS. While HRV may sound attractive as a biomarker of response, it would be premature to state that this alone could direct this field forward. Searching for a reliable biomarker for effective LLTS should be a continued focus of future research. The aforementioned limitations show clear paths for future research studies.
Conclusion
Non-invasive autonomic modulation using LLTS appears promising in its early stages. The ability to stimulate the central vagal complexes at a level below the heart-rate-lowering threshold seems very attractive. LLTS has promising data in animal models of AF, post-acute MI and heart failure. Accumulating data in humans with paroxysmal AF, post-acute MI and both systolic and diastolic heart failure may pave the way for larger clinical trials focused on demonstrating improvement in morbidity and mortality associated with such disease states. Larger trials are needed to prove the safety and efficacy of tragus stimulation.
Clinical Perspective
- Tragus stimulation is an emerging therapy for non-invasive modulation of the autonomic nervous system.
- Pre-clinical and clinical studies proved the potential of tragus stimulation in AF, MI and heart failure.
- Though the number of clinical trials is still limited, available data point to safety and efficacy of tragus stimulation. Large-scale trials are critically warranted.